A) q
B) qrev/T
C) qrev
D) Tqrev
E) q + w
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔS° for the decomposition of phosphorous trichloride into its constituent elements, 2PCl3 (g) → P2 (g) + 3Cl2( g) Is __________ J/K∙ mol.
A) -311.7
B) +311.7
C) +263.6
D) +129.4
E) -129.4
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate ΔG° for the autoionization of water at 25°C. Kw = 1.0 × 10-14
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following, the entropy of gaseous __________ is the largest at 25°C and 1 atm.
A) CH3OH
B) C2H5OH
C) C3H7OH
D) CH4
E) C4H10
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second law of thermodynamics states that __________.
A) ΔE = q + w
B) ![]()
C) for any spontaneous process, the entropy of the universe increases
D) the entropy of a pure crystalline substance is zero at absolute zero
E) ΔS = qrev/T at constant temperature
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following, only __________ is not a state function.
A) S
B) H
C) q
D) E
E) T
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The standard Gibbs free energy of formation of __________ is zero. (a) H2O (l) (b) O (g) (c) I2 (s)
A) (a) only
B) (b) only
C) (c) only
D) (b) and (c)
E) (a) , (b) , and (c)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔH° for the oxidation of solid elemental sulfur to gaseous sulfur dioxide, S (s, rhombic) + O2 (g) → SO2 (g) Is __________ kJ/mol.
A) +269.9
B) -269.9
C) +0.00
D) -11.6
E) +11.6
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction C(s) + H2O(g) → CO(g) + H2(g) ΔH° = 131.3 kJ/mol and ΔS° = 127.6 J/K ∙ mol at 298 K. At temperatures greater than __________°C this reaction is spontaneous under standard conditions.
A) 273
B) 325
C) 552
D) 756
E) 1029
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
ΔS is negative for the reaction __________.
A) 2SO2 (g) + O2 (g) → 2SO3 (g)
B) NH4Cl (s) → NH3 (g) + HCl (g)
C) PbCl2 (s) → Pb2+ (aq) + 2Cl- (aq)
D) 2C (s) + O2 (g) → 2CO2 (g)
E) H2O (l) → H2O (g)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Find the temperature (in K)above which a reaction with a ΔH of 123.0 kJ/mol and a ΔS of 90.00 J/K∙ mol becomes spontaneous.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: NH3 (g) + HCl (g) → NH4Cl (s)
Given the following table of thermodynamic data,  determine the temperature (in °C) above which the reaction is nonspontaneous.
determine the temperature (in °C) above which the reaction is nonspontaneous.
A) This reaction is spontaneous at all temperatures.
B) 618.1
C) 432.8
D) 345.0
E) 1235
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a system is at equilibrium, __________.
A) the reverse process is spontaneous but the forward process is not
B) the forward and the reverse processes are both spontaneous
C) the forward process is spontaneous but the reverse process is not
D) the process is not spontaneous in either direction
E) both forward and reverse processes have stopped
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a pure crystalline solid that is heated from absolute zero to a temperature above the boiling point of the liquid. Which of the following processes produces the greatest increase in the entropy of the substance?
A) melting the solid
B) heating the liquid
C) heating the gas
D) heating the solid
E) vaporizing the liquid
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔH° for the decomposition of POCl3 into its constituent elements, 2POCl3 (g) → P2 (g) + O2 (g) + 3Cl2 (g) Is __________ kJ/mol.
A) -1228.7
B) +1228.7
C) -940.1
D) +940.1
E) +0.00
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The normal boiling point of ethanol (C2H5OH) is 78.3°C and its molar enthalpy of vaporization is 38.56 kJ/mol. What is the change in entropy in the system in J/K when 97.2 grams of ethanol at 1 atm condenses to a liquid at the normal boiling point?
A) 330
B) 1038
C) -330
D) -1038
E) -231
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reaction produces an increase in the entropy of the system?
A) Ag+ (aq) + Cl- (aq) → AgCl (s)
B) CO2 (s) → CO2 (g)
C) H2 (g) + Cl2 (g) → 2HCl (g)
D) N2 (g) + 3H2 (g) → 2NH3 (g)
E) H2O (l) → H2O (s)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The value of ΔG° at 25°C for the following reaction: C2H4 (g) + H2 (g) → C2H6 (g) Is __________ kJ/mol. At 298 K, ΔH° for this reaction is -137.5 kJ/mol, and ΔS° is +120.5 J/K.
A) -35800
B) -173.4
C) 35800
D) -101.7
E) -274.2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Find the temperature (in K)above which a reaction with a ΔH of 53.00 kJ/mol and a ΔS of 100.0 J/K∙ mol becomes spontaneous.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction: FeO (s) + Fe (s) + O2 (g) → Fe2O3 (s)
Given the following table of thermodynamic data at 298 K: 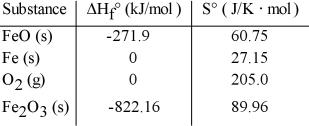 The value K for the reaction at 25°C is __________.
The value K for the reaction at 25°C is __________.
A) 370
B) 5.9 × 104
C) 3.8 × 10-14
D) 7.1 × 1085
E) 8.1 × 1019
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 120
Related Exams