A) It absorbs UV radiation and undergoes fragmentation.
B) It reflects UV radiation back into space.
C) It reflects heat back into space.
D) It emits UV radiation when excited by sunlight.
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bond energies increase in going from C-N (lowest) to C-O to C-F (highest) .Explain this trend based upon the atomic sizes of these atoms as deduced from their positions in the periodic table.
A) In going from nitrogen to oxygen to fluorine the atoms get larger.This means a greater nuclear charge,which translates into stronger chemical bonds.
B) In going from nitrogen to oxygen to fluorine the atoms get smaller.This means a greater nuclear charge,which translates into stronger chemical bonds.
C) In going from nitrogen to oxygen to fluorine the atoms get larger.This means that the bonding atoms are farther apart,which translates into a greater bond energy.
D) In going from nitrogen to oxygen to fluorine the atoms get smaller.This means that the bonding atoms are closer together,which translates into a greater bond energy.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following reaction energies is the least exothermic (but still an exothermic reaction) ?
A) 540 kJ/mole
B) -540 kJ/mole
C) 125 kJ/mole
D) -125 kJ/mole
E) not enough information given
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do the natural reactions involving ozone not lead to depletion of the ozone layer?
A) Ozone continually rises from the troposphere.
B) Ozone is too diffuse.
C) Ozone breaks into fragments that can reassemble into more ozone.
D) UV radiation generates more ozone.
E) c and d
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass of an oxygen atom,O,in atomic mass units?
A) 12 amu
B) 16 amu
C) 18 amu
D) 32 amu
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
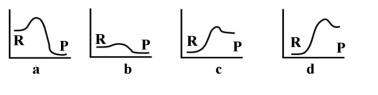 -For the above energy profiles,which reaction has the lowest activation energy?
-For the above energy profiles,which reaction has the lowest activation energy?
A) a
B) b
C) c
D) d
E) All have the same activation energy.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula mass of a molecule of CO2?
A) 44 amu
B) 56 amu
C) 58.9 amu
D) 118 amu
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 -For the above energy profiles,which reaction has the highest activation energy?
-For the above energy profiles,which reaction has the highest activation energy?
A) a
B) b
C) c
D) d
E) All have the same activation energy.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following balanced equation,which has the highest coefficient? 4 H2 + 2 C → 2 CH4
A) H2
B) C
C) CH4
D) H4
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many molecules of aspirin (formula mass aspirin = 180.0 amu) are there in a 0.250-gram sample?
A) 6.02 ×
B) 8.36 ×
C) 1.51 ×
D) More information is needed.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If it takes 20 beryllium atoms to equal the mass of two krypton atoms,what is the relative mass of beryllium compared to krypton?
A) 1/10
B) 1/20
C) 40 times
D) 100 times
E) 10 times
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An Alka-Seltzer antacid tablet bubbles vigorously when placed in water but only slowly when placed in an alcoholic beverage of the same temperature containing a 50:50 mix of alcohol and water.Propose a probable explanation involving the relationship between the speed of a reaction and molecular collisions.
A) The alcohol absorbs the carbon dioxide bubbles before they escape the liquid phase.
B) Alcohol molecules are more massive than water molecules,hence they move slower and their collisions are not as forceful.
C) The tablet reacts with water but not the alcohol.
D) In a 50:50 mix there are fewer water molecules for the antacid molecules to collide with.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The yeast in bread dough feeds on sugar to produce carbon dioxide.Why does the dough rise faster in a warmer area?
A) There is a greater number of effective collisions among reacting molecules.
B) Atmospheric pressure decreases with increasing temperature.
C) The yeast tends to "wake up" with warmer temperatures,which is why baker's yeast is best stored in the refrigerator.
D) The rate of evaporation increases with increasing temperature.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 1.00 carat pure diamond has a mass of 0.20 grams.How many carbon atoms are there within this diamond?
A) 6.0 × carbon atoms
B) 2.0 × carbon atoms
C) 1.0 × carbon atoms
D) 6.0 ×
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concept of entropy tells us how exothermic reactions are
A) favored over endothermic reactions.
B) not as favorable as endothermic reactions.
C) just as effective as endothermic reactions.
D) the reverse of endothermic reactions.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is greater: 1.01 amu of hydrogen or 1.01 grams of hydrogen?
A) 1.01 amu of hydrogen is greater than 1.01 grams of hydrogen.
B) 1.01 grams of hydrogen is greater than 1.01 amu of hydrogen.
C) 1.01 grams of hydrogen and 1.01 amu of hydrogen have the same mass.
D) Not enough information information is provided.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of water can be formed from the reaction between 10 grams of oxygen and 1 gram of hydrogen?
A) 11 grams of water are formed since mass must be conserved.
B) 10 grams of water are formed since you can't get a greater mass of water produced than oxygen reacting.
C) 9 grams of water are formed because oxygen and hydrogen react in an 8:1 ratio.
D) No water is formed because there is insufficient hydrogen to react with the oxygen.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which has the greatest number of atoms?
A) 28 g of nitrogen,
B) 32 g of oxygen,
C) 16 g of methane,C
D) 38 g of fluorine,
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Small samples of oxygen gas needed in the laboratory can be generated by any number of simple chemical reactions,such as 2 KCl (s) ? 2 KCl (s) + 3 (g) What mass of oxygen (in grams) is produced when 122.6 g of KCl (formula mass = 122.6 amu) takes part in this reaction?
A) 32.00 grams
B) 48.00 grams
C) 96.00 grams
D) More information is needed.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much energy,in kilojoules,is released or absorbed from the reaction of one mole of nitrogen, ,with three moles of molecular hydrogen, ,to form two moles of ammonia,N ? H-N (bond energy: 389 kJ/mol)
H-H (bond energy: 436 kJ/mol)
N  N (bond energy: 946 kJ/mol)
N
N (bond energy: 946 kJ/mol)
N  N + H-H + H-H ? N + N
N + H-H + H-H ? N + N
A) +899 kJ/mol absorbed
B) -993 kJ/mol released
C) +80 kJ/mol absorbed
D) -80 kj/mol released
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 120
Related Exams