A) 0.33
B) 0.0012
C) 0.0025
D) 0.67
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The figure shows a pV diagram for 0.0061 mol of ideal gas that undergoes the process 1 → 2 → 3.What is the pressure p2? (R = 8.31 J/mol ∙ K) 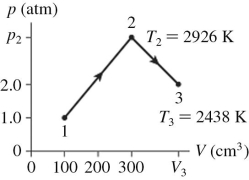
A) 4.9 atm
B) 4.9 × 105 atm
C) 15 atm
D) 1.5 × 106 atm
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The process shown on the pV diagram in the figure is 
A) adiabatic.
B) isothermal.
C) isochoric.
D) isobaric.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An important feature of the Carnot cycle is that
A) its efficiency can be 100%.
B) its efficiency depends only on the absolute temperature of the hot reservoir used.
C) its efficiency is determined by the temperatures of the hot and cold reservoirs between which it works and by the properties of the working substance used,and on nothing else.
D) it is an example of an irreversible process that can be analyzed exactly without approximations.
E) no engine can be more efficient than a Carnot engine operating between the same two temperatures.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain gas is compressed adiabatically.The amount of work done on the gas is 800 J.What is the change in the internal (thermal) energy of the gas?
A) 800 J
B) -800 J
C) 400 J
D) 0 J
E) More information is needed to answer this question.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During each cycle,the compressor in a certain ideal Carnot refrigerator performs 480 J of work to remove 150 J of heat from the interior of the refrigerator.How much heat do the coils behind the refrigerator discharge into the kitchen each cycle?
A) 110 J
B) 150 J
C) 330 J
D) 480 J
E) 630 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal reversible heat pump is taking heat from the outside air at -10.0°C and discharging it into the house at 18.0°C.What is the coefficient of performance of this heat pump?
A) 10.4
B) 9.44
C) 0.644
D) 0.533
E) 0.0962
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas is taken through the cycle shown in the pV diagram in the figure.During one cycle,how much work is done by the gas? 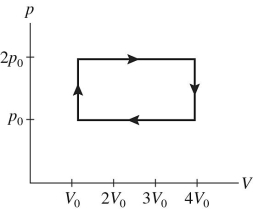
A) p0V0
B) 2 p0V0
C) 3 p0V0
D) 4 p0V0
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The process shown on the pV diagram in the figure is an 
A) adiabatic expansion.
B) isothermal expansion.
C) isometric expansion.
D) isobaric expansion.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal gas is compressed isobarically to one-third of its initial volume.The resulting pressure will be
A) three times as large as the initial value.
B) equal to the initial value.
C) more than three times as large as the initial value.
D) impossible to predict on the basis of this data.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 90 of 90
Related Exams