A) 2.0 M Ar
B) 1.0 M H2
C) 2.0 M N2
D) 0.5 M Ne
E) 2.0 M O2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume will a balloon occupy at 1.0 atm,if the balloon has a volume of 8.8 L at 4.4 atm?
A) 2.0 L
B) 0.50 L
C) 39 L
D) 13 L
E) 4.4 L
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of 0.200 moles of nitrogen occupies 0.400 L.Under the same conditions,what number of moles occupies 1.200 L?
A) 0.600 moles
B) 0.0667 moles
C) 2.40 moles
D) 0.0960 moles
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 55.0-L steel tank at 20.0°C contains acetylene gas,C2H2,at a pressure of 1.39 atm.Assuming ideal behavior,how many grams of acetylene are in the tank?
A) 3.17 g
B) 8.20 g
C) 82.9 g
D) 1210 g
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate of effusion of oxygen to an unknown gas is 0.935.What is the other gas?
A) Ne
B) Ar
C) F2
D) N2
F) B) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Give the gas that is the lowest percent by volume in dry gas.
A) CO2
B) He
C) H2
D) N2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 1.000 kg sample of nitroglycerine,C3H5N3O9,explodes and releases gases with a temperature of 1985°C at 1.100 atm.What is the volume of gas produced? 4 C3H5N3O9(s) → 12 CO2(g) + 10 H2O(g) + 6 N2(g) + O2(g)
A) 742.2 L
B) 3525 L
C) 4730 L
D) 5378 L
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Calcium hydride ( 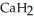 ) reacts with water to form hydrogen gas:
) reacts with water to form hydrogen gas: 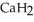 (s) +
(s) + 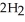 O(l) →
O(l) → 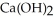 (aq) +
(aq) + 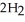 (g) How many grams of
(g) How many grams of 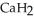 are needed to generate 48.0 L of
are needed to generate 48.0 L of 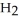 gas at a pressure of 0.888 atm and a temperature of 32°C?
gas at a pressure of 0.888 atm and a temperature of 32°C?
A) 50.7
B) 0.851
C) 143
D) 35.8
E) 71.7
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What pressure will 2.6 × 1023 molecules of N2 exert in a 3.9 L container at 45°C?
A) 5.7 atm
B) 1.7 atm
C) 2.9 atm
D) 3.4 atm
E) 4.6 atm
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas mixture contains CO,Ar and H2.What is the total pressure of the mixture,if the mole fraction of H2 is 0.350 and the pressure of H2 is 0.480 atm?
A) 1.37 atm
B) 0.168 atm
C) 5.95 atm
D) 0.729 atm
E) 2.1 atm
G) A) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following would have a density of 1.37 g/L at 7.0°C and 0.987 atm?
A) N2
B) O2
C) Kr
D) Rn
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.465 g sample of an unknown compound occupies 245 mL at 298 K and 1.22 atm.What is the molar mass of the unknown compound?
A) 26.3 g/mol
B) 33.9 g/mol
C) 12.2 g/mol
D) 38.0 g/mol
E) 81.8 g/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the volume of 0.700 mol of neon gas at a pressure of 609.5 mm Hg and 117°C?
A) 0.0941 L
B) 13.5 L
C) 2.13 L
D) 27.9 L
E) 0.138 L
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following gases in order of increasing density at STP. F2 NH3 N2O4 Ar
A) N2O4 < Ar < F2 < NH3
B) Ar < N2O4 < F2 < NH3
C) F2 < Ar < N2O4 < NH3
D) NH3 < F2 < Ar < N2O4
E) Ar < F2 < NH3 < N2O4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of nitrogen will react with 380 mL of hydrogen at -30°C and 200 mm Hg? N2(g) + 3 H2(g) → 2 NH3(g)
A) 0.0150 mol
B) 0.00782 mol
C) 0.00501 mol
D) 0.00167 mol
E) 0.0382 mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following samples will have the lowest pressure if they are all at the same temperature and in identical containers (same volume) ?
A) 15 g H2
B) 15 g Ar
C) 15 g Kr
D) 15 g CO2
E) All of these samples will have the same pressure.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gas occupies 4.23 L at 2.25 atm.What is the volume at 3.46 atm?
A) 6.50 L
B) 1.84 L
C) 2.75 L
D) 32.9 L
E) 0.364 L
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of decreasing rate of effusion. F2 SF6 He Ar
A) SF6 > Ar > F2 > He
B) Ar > He > SF6 > F2
C) F2 > Ar > He > SF6
D) He > F2 > Ar > SF6
E) He > F2 > SF6 > Ar
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Zinc reacts with aqueous sulfuric acid to form hydrogen gas: Zn(s) + 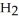
 (aq) → Zn
(aq) → Zn 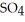 (aq) +
(aq) + 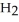 (g)
In an experiment,201 mL of wet
(g)
In an experiment,201 mL of wet 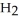 is collected over water at 27°C and a barometric pressure of
is collected over water at 27°C and a barometric pressure of 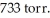 The vapor pressure of water at 27°C is 26.74 torr.The partial pressure of hydrogen in this experiment is ________ atm.
The vapor pressure of water at 27°C is 26.74 torr.The partial pressure of hydrogen in this experiment is ________ atm.
A) 0.929
B) 706
C) 0.964
D) 760
E) 1.00
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the gases according to increasing rate of diffusion at 50°C.
A) NO2 < O2 < SO2
B) O2 < NO2 < SO2
C) O2 < SO2 < NO2
D) SO2 < O2 < NO2
E) SO2 < NO2 < O2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 185
Related Exams