A) Substrate binds to an allosteric site rather than to the active site of an enzyme.
B) Binding of an activator molecule changes the shape of the active site of an enzyme.
C) The conformation of the active site is determined by the tertiary or quaternary structure of the enzyme.
D) Binding of substrate to the active site changes the shape of the active site of an enzyme.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aspects of enzyme structure is best described by a clasping handshake analogy?
A) the specific manner in which an enzyme folds to form secondary and tertiary structures
B) the specific manner in which an enzyme interacts with water
C) the specific manner in which an enzyme binds substrate
D) the specific manner in which an enzyme is denatured by low pH
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A decrease in entropy is associated with which type of reaction?
A) dehydration
B) catabolic
C) depolymerization
D) hydrolysis
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do hydrolysis reactions occur more readily in solution than dehydration reactions?
A) Hydrolysis reactions increase G, or Gibbs free energy of the system.
B) Hydrolysis reactions are endergonic and increase entropy of the system.
C) Hydrolysis reactions are exergonic and decrease entropy of the system.
D) Hydrolysis reactions are exergonic and increase entropy of the system.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true of metabolism in its entirety in all organisms?
A) Metabolism depends on a constant supply of energy from food.
B) Metabolism uses all of an organism's resources.
C) Metabolism consists of all the energy transformation reactions in an organism.
D) Metabolism manages the increase of entropy in an organism.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is a logical consequence of the second law of thermodynamics?
A) If the entropy of a system increases, there must be a corresponding decrease in the entropy of the universe.
B) If the entropy of a system decreases, there must be a corresponding decrease in the entropy of the universe.
C) If there is an increase in the energy of a system, there must be a corresponding decrease in the energy of the rest of the universe.
D) Each chemical reaction in an organism must increase the total entropy of the universe.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is consistent with the second law of thermodynamics?
A) A gain of free energy in a system is always associated with conversion of energy from one form to another.
B) A constant input of energy is required to maintain the high level of cellular organization.
C) Without an input of energy, the entropy of an organism would tend to decrease over time.
D) Every energy transformation performed by an organism decreases the entropy of the universe.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How does a noncompetitive inhibitor decrease the rate of an enzyme-catalyzed reaction?
A) by binding to the active site of the enzyme, thus preventing binding of the normal substrate
B) by binding to an allosteric site, thus changing the shape of the active site of the enzyme
C) by decreasing the free-energy change of the reaction catalyzed by the enzyme
D) by binding to the substrate, thus changing its shape so that it no longer binds to the active site of the enzyme
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How might a change of one amino acid at a site, distant from the active site of an enzyme, alter the substrate specificity of an enzyme?
A) by changing the stability of the enzyme
B) by changing the three-dimensional conformation of the enzyme
C) by changing the optimum pH for the enzyme
D) by changing the binding site for a noncompetitive inhibitor
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Zinc, an essential trace element for most organisms, is present in the active site of the enzyme carboxypeptidase. The zinc most likely functions as ________.
A) a noncompetitive inhibitor of the enzyme
B) an allosteric activator of the enzyme
C) a cofactor necessary for enzyme activity
D) a coenzyme derived from a vitamin
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements describes an example of cooperativity associated with enzyme regulation?
A) binding of the end product of a metabolic pathway to the first enzyme in the pathway to inhibit the enzyme
B) one enzyme in a metabolic pathway passing its product to act as a substrate for the next enzyme in the pathway
C) binding a substrate to one subunit of a tetramer stimulates faster binding of substrate to each of the other three subunits
D) binding of an ATP molecule along with another substrate molecule in the active site of the enzyme
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
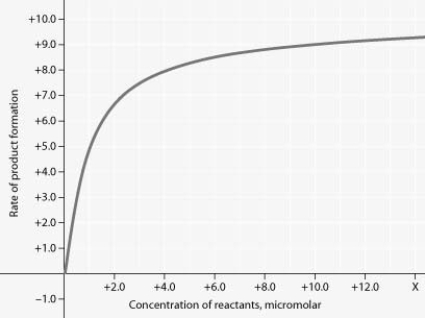 Rate of an enzyme-catalyzed reaction as a function of varying reactant concentration, with the concentration of enzyme constant.
For the enzyme-catalyzed reaction shown in the figure, if the initial reactant concentration is 1.0 micromolar, which of these treatments will cause the greatest increase in the rate of the reaction?
Rate of an enzyme-catalyzed reaction as a function of varying reactant concentration, with the concentration of enzyme constant.
For the enzyme-catalyzed reaction shown in the figure, if the initial reactant concentration is 1.0 micromolar, which of these treatments will cause the greatest increase in the rate of the reaction?
A) doubling the activation energy needed
B) cooling the reaction by 10°C
C) doubling the enzyme concentration
D) increasing the concentration of reactants to 10.0 micromolar, while reducing the concentration of enzyme by 1/2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following information to answer the question below.
(a)
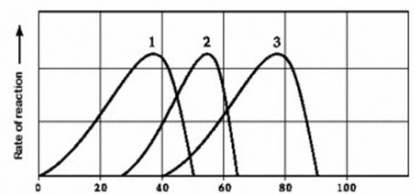 (b)
(b)
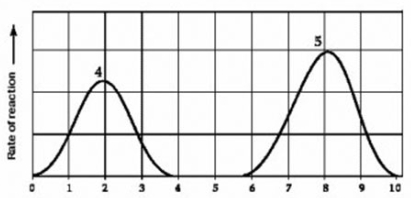 Activity of various enzymes at various temperatures (a) and at various pH (b)
Which curves on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
Activity of various enzymes at various temperatures (a) and at various pH (b)
Which curves on the graphs may represent the temperature and pH profiles of an enzyme taken from a bacterium that lives in a mildly alkaline hot springs at temperatures of 70°C or higher?
A) curves 1 and 5
B) curves 2 and 5
C) curves 3 and 4
D) curves 3 and 5
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Biological systems use free energy based on empirical data that all organisms require a constant energy input. The first law of thermodynamics states that energy can be neither created nor destroyed. For living organisms, which of the following statements is an important consequence of this first law?
A) The energy content of an organism is constant except for when its cells are dividing.
B) The organism must ultimately obtain all the necessary energy for life from its environment.
C) The entropy of an organism decreases with time as the organism grows in complexity.
D) Organisms are unable to transform energy from the different states in which it can exist.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following information to answer the question below.
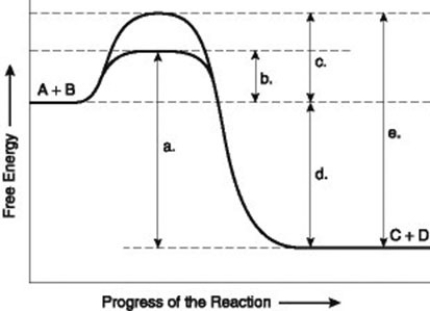 The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following in the figure would be the same in either an enzyme-catalyzed or a noncatalyzed reaction?
The figure illustrates the energy states associated with the reaction A + B ↔ C + D. Which of the following in the figure would be the same in either an enzyme-catalyzed or a noncatalyzed reaction?
A) a
B) b
C) c
D) d
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Protein kinases are enzymes that transfer the terminal phosphate from ATP to an amino acid residue on the target protein. Many are located on the plasma membrane as integral membrane proteins or peripheral membrane proteins. What purpose may be served by their plasma membrane localization?
A) ATP is more abundant near the plasma membrane.
B) They can more readily encounter and phosphorylate other membrane proteins.
C) Membrane localization lowers the activation energy of the phosphorylation reaction.
D) They flip back and forth across the membrane to access target proteins on either side.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If an enzyme is added to a solution where its substrate and product are in equilibrium, what will occur?
A) Additional substrate will be formed.
B) The reaction will change from endergonic to exergonic.
C) The free energy of the system will change.
D) Nothing; the reaction will stay at equilibrium.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true when comparing an uncatalyzed reaction to the same reaction with a catalyst?
A) The catalyzed reaction will be slower.
B) The catalyzed reaction will have the same ∆G.
C) The catalyzed reaction will have higher activation energy.
D) The catalyzed reaction will consume all of the catalyst.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If an enzyme in solution is saturated with substrate, the most effective way to obtain a faster yield of products is to
A) add more of the enzyme.
B) heat the solution to 90°C.
C) add more substrate.
D) add a noncompetitive inhibitor.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules is most similar in structure to ATP?
A) a pentose sugar
B) a DNA nucleotide
C) an RNA nucleotide
D) an amino acid with three phosphate groups attached
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 67
Related Exams